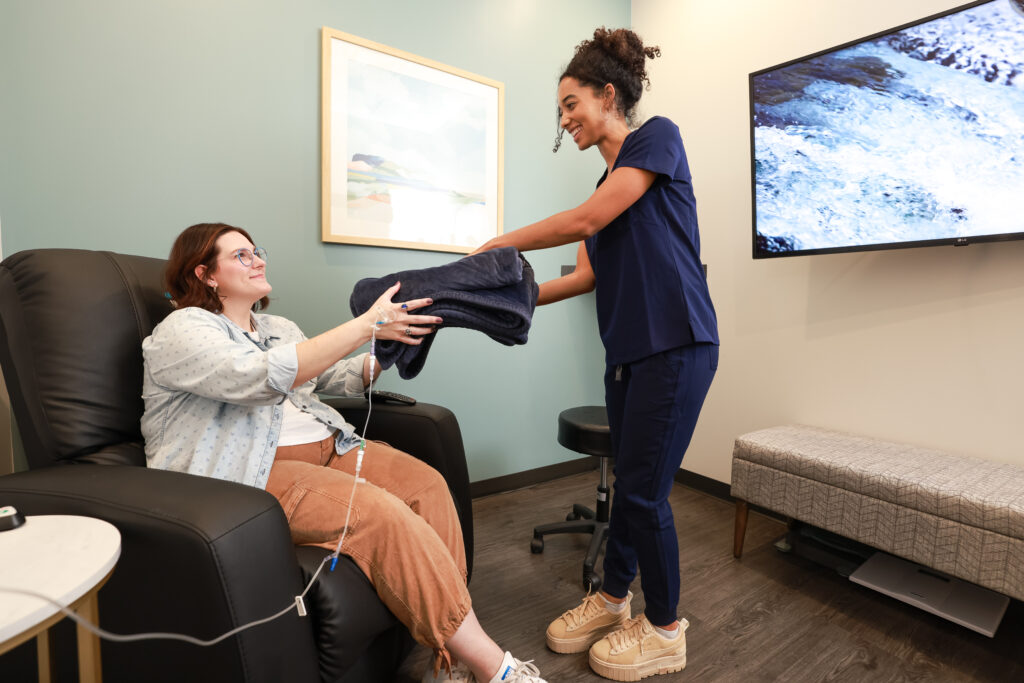
See why thousands of patients choose our centers as their preferred destination for ongoing care, including Skyrizi IV Infusion treatments.
Important Announcement
On June 17, 2022, the pharmaceutical manufacturer AbbVie announced that its product Skyrizi® received FDA approval for the treatment of moderately to severely active Crohn’s disease in adults.
The prescribing information for Skyrizi for the treatment of Crohn’s disease includes the administration of three intravenous infusion treatments over an 8-week period, followed by the ongoing self-administration of a subcutaneous injection. The three initial infusions must be administered by a healthcare professional, and IVX Health is now accepting Skyrizi IV referrals.
NOTE: Both the IV infusion formulation and indication for Crohn’s disease were recently approved by the FDA, and many healthcare payors have yet to officially recognize the new formulation and indication in their health insurance plans. IVX’s ability to receive a prior authorization for treatment may be temporarily delayed.
IVX Health is actively communicating with each of the healthcare payors and will proactively update both patients and referring providers as the issue is resolved.
At IVX, patients are our number one priority. If you have any additional questions, please contact your local IVX Health center immediately to discuss next steps.
Skyrizi IV is a biologic therapy that is indicated to treat:
- adults with moderate to severe Crohn’s disease
What is Skyrizi IV?
Skyrizi IV (risankizumab-rzaa) is a prescription medication administered via IV infusion used to treat adult individuals that have Crohn’s disease.
What is Skyrizi IV Used For?
Skyrizi IV is a prescription medicine used to treat adults with moderate to severe Crohn’s disease.
How to Get a Referral for Skyrizi IV
To receive a Skyrizi (risankizumab-rzaa) infusion at IVX Health, the patient must provide their referring physician’s order.
Patient information should include:
- Name
- Current address
- Home and mobile phone number
- Insurance information
Information the physician must include on the patient’s order for Sykrizi infusion therapy:
- Patient’s name and DOB
- Order date
- Diagnosis
- Physician’s name and NPI#
- Office address, phone and fax numbers
- Provider’s signature
Supporting clinical documentation from the physician should include:
- Result of Tuberculosis (TB) skin/ lab testing
- Baseline Liver Enzymes and Bilirubin
At IVX Health, patients receive high-quality treatment from compassionate medical professionals. If you have Crohn’s disease and are interested in receiving Skyrizi infusions at IVX Health, you can find your closest center by clicking here. Once you have your prescription, you can request an appointment.
Patients chose IVX Health to receive their ongoing Skyrizi IV treatments because:
- ALL private suites
- Comfortable recliners, with chairs for guests
- Evening & weekend appointments
- Friendly clinical staff deliver personal service
- Big screen TVs, high-speed WiFi, free snacks
Interested in moving your care to IVX Health?
Skyrizi IV Financial Assistance Programs
Information about financial assistance programs coming soon.
Skyrizi IV Related Blog Posts
Interested?
IVX Health would love to learn more about your care needs. If you’re interested in switching your infusion services – or if you just want to learn more – let us know.